A brain tumor is an abnormal growth of tissue in the brain. Brain tumors may develop from neural elements within the brain (primary brain tumor), or they may represent spread of distant cancers (secondary brain tumor). According to the National Cancer Institute, in 2013 the estimated new cases and deaths from brain tumor in the United States would be 23,130 and 14,080 respectively. There are many types of primary brain tumors. Primary brain tumors are named according to the type of cells or the part of the brain in which they originate. For example, most primary brain tumors begin in glial cells. This type of tumor is called a glioma. Among adults, the most common types are:
- Meningioma: This tumor arises in the meninges. It’s usually benign (grade I) and grows slowly.
- Oligodendroglioma: This tumor arises from cells that make the fatty substance that covers and protects nerves. It usually occurs in the cerebrum. It’s most common in middle-aged adults.
- Astrocytoma: This tumor arises from star-shaped glial cells called astrocytes.
Among these, glioblastoma multiforme (GBM) is the most common and deadliest of malignant primary brain tumors in adults. Classified as a Grade IV (most serious) astrocytoma, GBM develops from the lineage of star-shaped glial cells, called astrocytes that support nerve cells. Even though GBM is one of the most aggressive forms of human cancers, the incident of this disease is fairly less. Every year, in the United States approximately 7 out of 100,000 people are diagnosed with high-grade glioblastoma. Uncontrolled cellular proliferation, diffuse infiltration, and resistance to apoptosis are considered as hallmarks of GBM. The current standard of treatment in glioblastoma involves surgery followed by radiation therapy, and chemotherapy with the DNA alkylating agent temozolomide (TMZ). The efficacy of this current treatment strategy is limited as the median survival is just over one year because of development of resistance. Therefore, more effective treatment plans need to be developed and explored. In a recent study published in the peer review journal Clinical Cancer Research (2013 Apr 25) Thorpe and colleagues reported that combined treatment of ataxia telangiectasia mutated (ATM) inhibitor and radiation significantly increased survival of mice bearing glioblastoma with p53 mutation compared to treatment with ATM inhibitor or radiation alone.
The nuclear protein kinase ataxia-telangiectasia mutated (ATM) plays a crucial role in DNA-damage response by transducing double-strand breaks. This kinase phosphorylates several effectors that play key roles in cell cycle progression and DNA repair pathways.
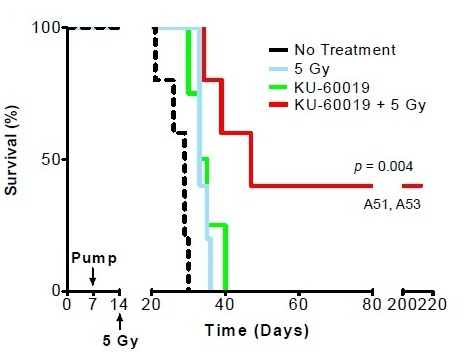
Ionizing radiation is the most effective therapy for glioblastoma. Ionizing radiation works by damaging the DNA of exposed tissue leading to cellular death. However, tumor cells evade death and contribute to radioresistance through preferential activation of the DNA damage checkpoint response and an increase in DNA repair capacity. Therefore, combined treatment of ionizing radiation with inhibitors of DNA damage checkpoint may overcome the problem of resistance in glioblastoma. Since ATM is associated with DNA damage repair there are several small molecule ATM inhibitors that disrupt ATM function and enhances sensitivity of tumor cells to radiation as well as chemotherapeutic agents. ATM inhibitor KU-60019 is a potent inhibitor effectively blocking activation of key ATM targets and radiosensitizing human glioma cells in vitro. In their study Thorpe et al. (2013) showed that mice bearing glioblastoma survived longer periods of time (145 days) when treated with KU-60019 and radiation than mice treated with radiation alone (58 days) or KU-60019 alone (66 days). In addition, this study also noted that mice bearing glioma with mutant p53 are more sensitive to KU-60019 dependent radiosensitization than mice bearing glioma with p53 wild-type. Altogether this study suggests that ATM kinase inhibition may be an effective strategy as adjuvant therapy for patients with mutant p53 brain cancers.
References:
1.Bao S, Wu Q, McLendon RE, Hao Y, Shi Q, Hjelmeland AB, Dewhirst MW, Bigner DD, Rich JN: Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature 2006, 444(7120):756-760.
2.Biddlestone-Thorpe L, Sajjad M, Rosenberg E, Beckta JM, Valerie NC, Tokarz M, Adams BR, Wagner AF, Khalil A, Gilfor D et al: ATM kinase inhibition preferentially sensitizes p53 mutant glioma to ionizing radiation. Clin Cancer Res 2013.
3. http://www.cancer.gov/cancertopics/wyntk/brain/page2