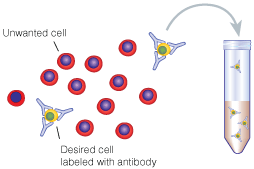
In a previous post, I covered the current options for isolating pure cell populations. One immediate question you will have to ask yourself is whether you would prefer positive selection or negative selection (depletion) for the isolation of your cell type of interest.
Positive selection involves the isolation of a target cell population by using an antibody that specifically binds that population. As an example, a positive selection kit for T cells would use an antibody specific for the CD3 molecule on T cells. Negative selection, however, involves the depletion of all cell types except your cell type of interest. With our T cell isolation example, our negative selection kit would likely involve antibodies specific for B cells (CD19), monocytes (CD14), NK cells (CD56), and so on. With the depletion of these cell types we would only be left with our cells of interest, in this case T cells (CD3).
The Advantages of Positive Selection
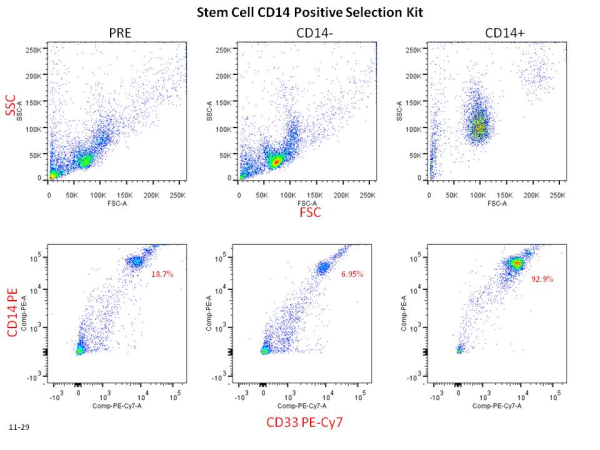
Positive selection and negative selection each have their advantages. Positive selection offers greater purity due to the specificity of the reaction. You know in our example that positive selection of T cells will only yield a high purity of T cells due to the binding of selection antibodies to CD3 molecules. Negative selection, however, is inherently leakier since it is impossible to design a perfect depletion cocktail to target all cells that do not carry CD3 molecules. It is important to point out though that all of the popular cell isolation companies have made quite excellent kits that yield good purity levels when done properly. The difference in purity between positive selection and negative selection is roughly 99% to 95% pure, both of which are more than serviceable.
their advantages. Positive selection offers greater purity due to the specificity of the reaction. You know in our example that positive selection of T cells will only yield a high purity of T cells due to the binding of selection antibodies to CD3 molecules. Negative selection, however, is inherently leakier since it is impossible to design a perfect depletion cocktail to target all cells that do not carry CD3 molecules. It is important to point out though that all of the popular cell isolation companies have made quite excellent kits that yield good purity levels when done properly. The difference in purity between positive selection and negative selection is roughly 99% to 95% pure, both of which are more than serviceable.
Another advantage of positive selection is that it offers the ability for a follow-up selection, or sequential isolations. Since negative selection works by binding all cells except the target cells with bead-bound antibodies, there is no way to do further isolations with the negative population. However, the negative flow through population from positive isolation will not have bead-bound antibodies and therefore is available for either another positive selection or a negative selection of your choice.
The Advantages of Negative Selection
The disadvantage of positive selection of course is that your isolated cells will carry bead-bound antibodies. Not surprisingly, the kit manufacturers will tell you that this is not a concern, but it is something you need to keep and mind and use at your discretion. While neither the antibodies nor the beads should activate your isolated cells, it may in some way affect your downstream experiments. If you feel this could be an issue and you would prefer ‘untouched’ cells, then negative selection may be the right choice for you. First, however, be sure the negative selection kit actually depletes all necessary cells in order to achieve a pure target population. Often these kits are designed for common target tissues, such as peripheral bloods, lymph nodes, and spleens. Unfortunately negative selection kits may not work well for other target tissues. For example, my own work involves isolation of T cells from tumor samples. Since stock negative selection kits do not contain depletion antibodies for tumor cells, negative selection is not an option for our assays, and as a result we are forced to use positive selection.
that your isolated cells will carry bead-bound antibodies. Not surprisingly, the kit manufacturers will tell you that this is not a concern, but it is something you need to keep and mind and use at your discretion. While neither the antibodies nor the beads should activate your isolated cells, it may in some way affect your downstream experiments. If you feel this could be an issue and you would prefer ‘untouched’ cells, then negative selection may be the right choice for you. First, however, be sure the negative selection kit actually depletes all necessary cells in order to achieve a pure target population. Often these kits are designed for common target tissues, such as peripheral bloods, lymph nodes, and spleens. Unfortunately negative selection kits may not work well for other target tissues. For example, my own work involves isolation of T cells from tumor samples. Since stock negative selection kits do not contain depletion antibodies for tumor cells, negative selection is not an option for our assays, and as a result we are forced to use positive selection.
It is important to choose an optimal cell isolation strategy specific to your assay, your target cells, and your tissue source. In my next post I will offer some tips for sorting through the various kits and technologies many companies offer for cell isolation.





 Colt Egelston is currently a post-doctoral fellow at the Beckman Research Institute of the City of Hope, in Duarte, CA. He received his Ph.D. from Rush University in Chicago and is interested in all things immunology.
Colt Egelston is currently a post-doctoral fellow at the Beckman Research Institute of the City of Hope, in Duarte, CA. He received his Ph.D. from Rush University in Chicago and is interested in all things immunology.
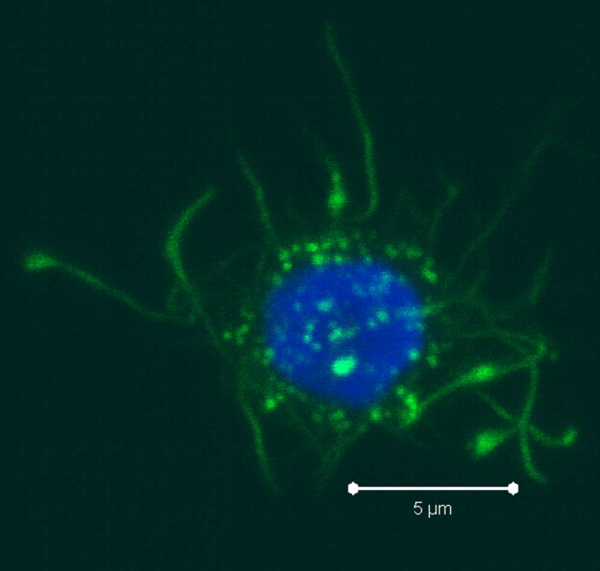
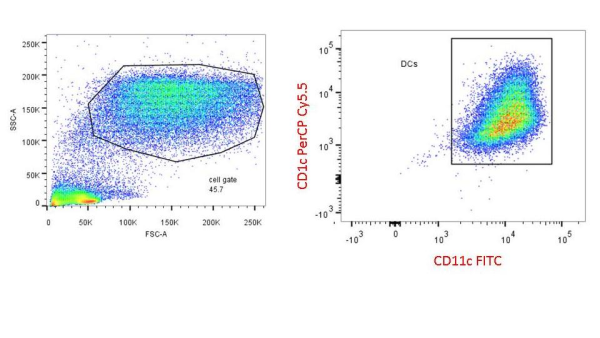
 PBMCs are not just a source of many different circulating immune cell types, but also a source of potential cells that one can generate in vitro. One excellent and long-standing example of this is the generation of dendritic cells (DCs) from monocytes. Monocyte derived DCs (mDCs) are an excellent tool for researchers to do immunological assays requiring a source of professional antigen presenting cells (APCs). While circulating B cells are capable of antigen presentation and T cell activation, they do not offer the robust response that DCs do. The generation of mDCs is a relatively simple protocol that anyone can do with just a source of PBMCs, a few important cytokines, and, of course, some media and incubator space. After this protocol, you will have obtained immature mDCs that can then be matured for use as APCs in your assay.
PBMCs are not just a source of many different circulating immune cell types, but also a source of potential cells that one can generate in vitro. One excellent and long-standing example of this is the generation of dendritic cells (DCs) from monocytes. Monocyte derived DCs (mDCs) are an excellent tool for researchers to do immunological assays requiring a source of professional antigen presenting cells (APCs). While circulating B cells are capable of antigen presentation and T cell activation, they do not offer the robust response that DCs do. The generation of mDCs is a relatively simple protocol that anyone can do with just a source of PBMCs, a few important cytokines, and, of course, some media and incubator space. After this protocol, you will have obtained immature mDCs that can then be matured for use as APCs in your assay. Once the culture period has finished, between 6-8 days, the mDCs can be collected. The exact day is not critical, as long as you remain consistent in the day you pick for your following experiments. To collect the mDCs, gently wash the culture dishes with several streams of media by pipetting up and down. The mDCs, which are currently immature, will be somewhat floating and only loosely adherent. Because of their loose adherence, they require several rounds of gentle pipetting, but do not require cell scraping, EDTA, or trypsin treatment. Note that the culture dishes will still contain some adherent cells. Do not worry about these cells, since these are not the loosely adherent DCs we are interested in.
Once the culture period has finished, between 6-8 days, the mDCs can be collected. The exact day is not critical, as long as you remain consistent in the day you pick for your following experiments. To collect the mDCs, gently wash the culture dishes with several streams of media by pipetting up and down. The mDCs, which are currently immature, will be somewhat floating and only loosely adherent. Because of their loose adherence, they require several rounds of gentle pipetting, but do not require cell scraping, EDTA, or trypsin treatment. Note that the culture dishes will still contain some adherent cells. Do not worry about these cells, since these are not the loosely adherent DCs we are interested in. Colt Egelston is currently a post-doctoral fellow at the Beckman Research Institute of the City of Hope, in Duarte, CA. He received his Ph.D. from Rush University in Chicago and is interested in all things immunology.
Colt Egelston is currently a post-doctoral fellow at the Beckman Research Institute of the City of Hope, in Duarte, CA. He received his Ph.D. from Rush University in Chicago and is interested in all things immunology.
