Human peripheral blood mononuclear cells (PBMC) are composed of heterogeneous populations of various immune cell types. CD4+ and CD8+ T cells are known to exist in various functional and differentiated states. Following antigen experience, naïve T cells are thought to progressively differentiate along a path through central memory, effector memory, and terminally differentiated effector states. Markers for differentiating PBMC T cells into these subtypes using multiparametric flow cytometry include: CD3, CD4, CD8, CD45RA or CD45RO, and CCR7 or CD62L.
The cytokine milieu T cells are exposed to during antigen encounter directs differentiation into various subtypes that exhibit unique functional properties and gene expression programs including cytokines, transcription factors, and surface markers. This is true for both CD4+ and CD8+ T cells. In a previous post, I discussed various markers that can be utilized by flow cytometry to identify CD4+ TH1, TH2, and TH17 populations in human PBMC.
CD8+ T cells can also differentiate into unique subsets similar to TH1, TH2, and TH17 CD4+ T cells. The CD8+ versions of these subsets are referred to as TC1, TC2, and TC17 CD8+ T cells, respectively, and are defined by expression of the same characteristic cytokines as their CD4+ counterparts.
As previously discussed, expression of subset-specific surface markers is easily determined by flow cytometry. Identification of intracellular cytokine production in T cells can be assessed following 4-6 hours of TCR stimulation with anti-CD3 and anti-CD28 antibodies or the combination of Phorbol 12-Myristate 13-Acetate (PMA) and ionomycin in the presence of brefeldin A or monensin. The cells are then fixed and permeabilized with buffers such as BD Biosciences’ Cytofix Cytoperm buffer set followed by antibody staining for cytokine expression and flow cytometry.
Like CD4+ TH1 cells, CD8+ TC1 cells characteristically produce IFNγ. This population is by far the most common cytokine-producing CD8+ cell subset, and is very easy to identify using intracellular staining for IFNγ.
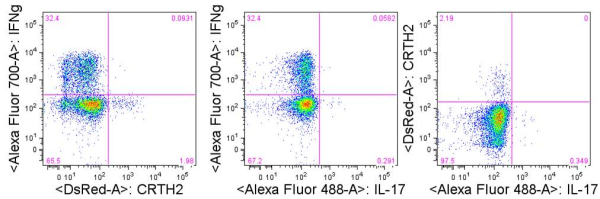
As with CD4+ TH2 cells, CD8+ TC2 cells can be identified by expression of IL-4, IL-5, and IL-13. Cosmi et. al, found that the surface marker CRTH2 was a robust marker for identification of CD8+ and CD4+ cells producing IL-4, IL-5, and IL-13 expression but not IFNγ. Expression of chemokine receptors CCR3 and CCR4 however, did not exclude IFNγ-producing cells. Because expression of IL-4, IL-5, and IL-13 can be difficult to detect, CRTH2 may be the easiest of these markers for TC2 identification in human PBMC.
CD8+ TC17 cells are characterized by expression of the cytokine IL-17. Expression of the chemokine receptors CCR5 and CCR6 were shown to enrich for IL-17 producing CD8+ cells. However CCR5 and CCR6 expression are also associated with TC1 cells, and thus may not be useful to differentiate between these subsets.

Figure: Expression of IFNγ, IL-17, and CRTH2 in CD8+ T cells from PBMC stimulated with for 4 hours with PMA/ionomycin.
In my own studies, I have utilized TCR or PMA/ionomycin stimulation of PBMCs to successfully identify IFNγ (TC1) and IL-17 (TC17) expressing cells, and CRTH2 expression to identify TC2 cells, as these may be the most robust markers for identification of these unique CD8+ T cell populations. Note also that these same markers reliably detect CD4+ TH1, TH17, and TH2 cells, respectively. Thus, these markers are useful to quantitate and study the function of both CD8+ and CD4+ subsets in human PBMC.
Additional Reading
Generation of polarized antigen-specific CD8 effector populations: reciprocal action of interleukin (IL)-4 and IL-12 in promoting type 2 versus type 1 cytokine profiles. Croft M, Carter L, Swain SL, Dutton RW. J Exp Med. 1994 Nov 1;180(5):1715-28.
CRTH2 is the most reliable marker for the detection of circulating human type 2 Th and type 2 T cytotoxic cells in health and disease. Cosmi L, Annunziato F, Galli MIG, Maggi RME, Nagata K, Romagnani S. Eur J Immunol. 2000 Oct;30(10):2972-9.
Cutting edge: Phenotypic characterization and differentiation of human CD8+ T cells producing IL-17. Kondo T, Takata H, Matsuki F, Takiguchi M. J Immunol. 2009 Feb 15;182(4):1794-8.
Functional expression of chemokine receptor CCR6 on human effector memory CD8+ T cells. Kondo T, Takata H, Takiguchi M. Eur J Immunol. 2007 Jan;37(1):54-65.