Malignant gliomas are classified by the World Health Organization (WHO), as grade IV tumors of neuroepithelial tissue and are the most common and deadly intracranial tumors, accounting for more than 70% of all brain tumors. One of the most prominent intrinsic behaviors of Glioblastoma Multiforme (GBM) is its invasiveness within the host’s central nervous system (CNS), in which single glioma cells travel a distance from the tumor mass and invade adjacent brain tissue
Due to the exceptional migratory ability of glioma cells, surgical resection of the tumor is almost always followed by tumor recurrence with foci located as close as 1 centimeter from the resection cavity or as far as the contralateral hemisphere. Despite advancements in surgical techniques and post-operational delivery of chemotherapeutics and radiation, the prognosis for glioma patients remains dismal making these lethal tumors virtually incurable. Although the history of glioma treatment dates back to the 19th century, the median survival of patients remains less than 14 months post-diagnosis. Thus, the only way to cure GBM is by essentially eliminating all glioma cells, including the single cells which have disseminated within the parenchyma, away from the tumor mass.
A promising potential strategy to treat high-grade brain tumors, is through cell-based therapies (CBTs) that incorporate autonomous tracking of tumor cells. Two examples of CBTs, which are currently under investigation in brain tumor clinical trials, include: 1) employing of genetically engineered neural stem cells (NSCs) as target-specific therapeutic-agent delivery vehicles, and 2) the adoptive transfer of tumor-specific, genetically engineered cytotoxic T-lymphocytes (CTLs).
The significance of NSCs and CTLs for treating CNS diseases:
One anatomical feature unique to the CNS is the presence of the blood brain barrier (BBB), which restricts the access of many compounds including many chemotherapeutic agents into the CNS. Thus, the BBB prevents effective drug delivery from the circulatory system to the tumor sites within the brain. Even when the drug is administered intracranially to overcome the limitations presented by the BBB, the densely packed environment of brain’s parenchyma inhibits effective diffusion of the drug throughout the brain and prevents the drug from reaching the tumor cells. These anatomical properties of the CNS are also responsible for inefficient distribution of gene therapy in the brain.
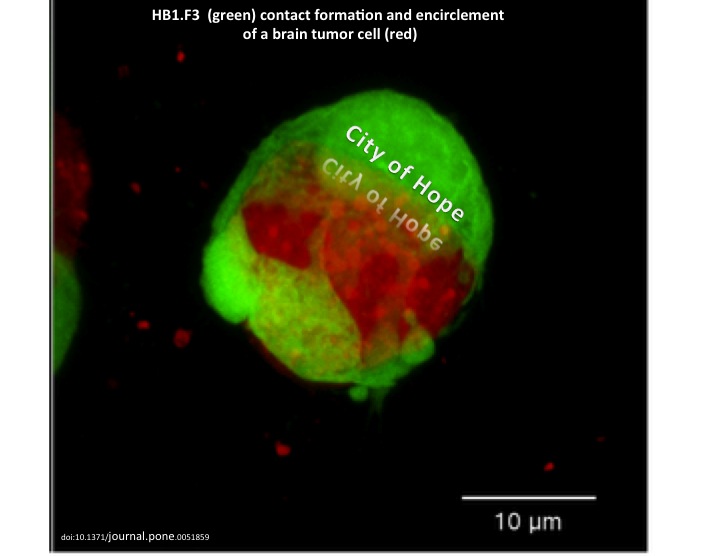
NSCs readily cross the BBB and intrinsically target invasive tumor cells that have migrated away from the tumor mass in vivo. The HB1.F3 NSC line, developed by Dr. Karen Aboody, is one clonally derived human cell line that is particularly well characterized and is used clinically for glioma therapy. Another advantage of this therapeutic model is its efficacy through both intracranial and intravenous administration, without rejection elicited by the host’s immune system against the NSCs; this is due to HB1.F3 NSCs’ low levels of MHC Class I antigen expression and undetectable levels of MHC Class II antigens.
Previous studies have indicated CTLs’ ability to target and kill GBM, medulloblastomas and therapeutically resistant subpopulations of glioma stem–like cancer-initiating cells (GSC), which express interleukin-13 receptor α2 (IL13Rα2). Although the BBB is not permeable to CTLs, Dr. Christine E. Brown reported a non-toxic strategy of delivering these T-cells to the CNS tumors, by placing a fibrin matrix-embedded with CTLs, in the resection cavity during surgery.

In vivo,secretion of monocyte chemotactic protein-1 (MCP-1) also known as chemokine C-C motif ligand 2 (CCL2), by the cancer cells attract CD4+ and CD8+ T-cells, leading to the subsequent host antitumor immune response. Brown’s group has shown that the same tumor-secreted chemoattractants will recruit genetically engineered CTLs. Since CTLs migrate freely within fibrin matrices, the presence of MCP-1 in the surrounding environment attracts the CTLs to migrate out of the fibrin. In their in vitro model, the IL13Rα2-specific T-cells successfully migrated out of the fibrin clot and killed the surrounding glioma cells.
Utilization of fibrin matrices allows a safe, non-toxic delivery of CTLs, without causing additional injury (i.e. injury caused by a catheter or needle injection) and inflammation to the brain tissue.
There are other therapeutic approaches for treatment of malignant brain tumors. Following are a list of further readings on the content of this article, as well as other current cancer research studies in regards to GBM:
Aboody, K., et al. “Translating stem cell studies to the clinic for CNS repair: current state of the art and the need for a Rosetta stone.” Neuron, 26 May 2011 (Vol. 70, Issue 4, pp. 597-613)