Myeloid suppressor cells (MSCs) such as of macrophages and myeloid derived suppressor cells (MDSCs) are thought to be key players in cancer promotion and resistance to therapy. MSCs originate from the myeloid lineage in the bone marrow and circulate the bloodstream. They are recruited from the peripheral blood to tissues or tumor sites by cytokines such as colony stimulating factor-1 (CSF-1) where they can differentiate into macrophages or MDSCs. Their normal role is to protect a host from possible autoimmune reactions and to subdue over active immune responses.
Macrophages can exist in various activation states depending on the cues they receive from their environment. Classically activated or M1 macrophages are said to be anti-tumor and pro-inflammatory. Signaling from cytokines such as GM-CSF, TNF, IFN-γ, or microbial stimuli such as LPS promote an M1 response. M1 macrophages are said to be cytotoxic, and secrete reactive oxygen species (ROS), and thus can damage tissue. Alternatively activated or M2 macrophages are said to be pro-tumorigenic and anti-inflammatory. Cytokines such as IL4, IL-13 and IL-10 are said to promote an M2 activation state, although the designation of IL-10 as an M2 skewing cytokines remains controversial in the field. M2 macrophages downregulate T-cell activity, secrete high levels of growth factors, angiogenenic factors and matrix remodeling enzymes. Thus, macrophages can either promote an anti-tumor inflammatory response or suppress it depending on the cytokines (signals) and they encounter.
The different activation states of macrophages can be characterized by variations in cell surface and intracellular marker expression levels. Both M1 and M2 macrophages express myeloid markers CD11b and CD33, monocyte marker CD14 as well as macrophage marker, glycoprotein CD68. M1 macrophages express high levels of pro-inflammatory cytokines IL-12, IL-23, and low levels of the anti-inflammatory cytokine IL-10 while the reverse is true for M2 macrophages. The mannose receptor CD206 and scavenger receptor CD163 are expressed at elevated levels in M2 macrophages. CD68, CD163 and CD206 markers are used largely for immunohistological characterization of macrophages although flow cytometry analysis with these marker is also possible.
Retrospective studies have been carried out on breast, melanoma, pancreatic, and non-small cell lung cancer specimens to name a few, assessing the phenotype of tumor associated macrophages based CD68, CD163, and CD206 expression levels. In general, a negative or poor prognosis was associated with higher levels of M2 versus M1 macrophages.
It is important to note that variations of the M2 phenotype exist. Additionally, these phenotypes are quite plastic and it is therefore possible for macrophages to switch between activation states.
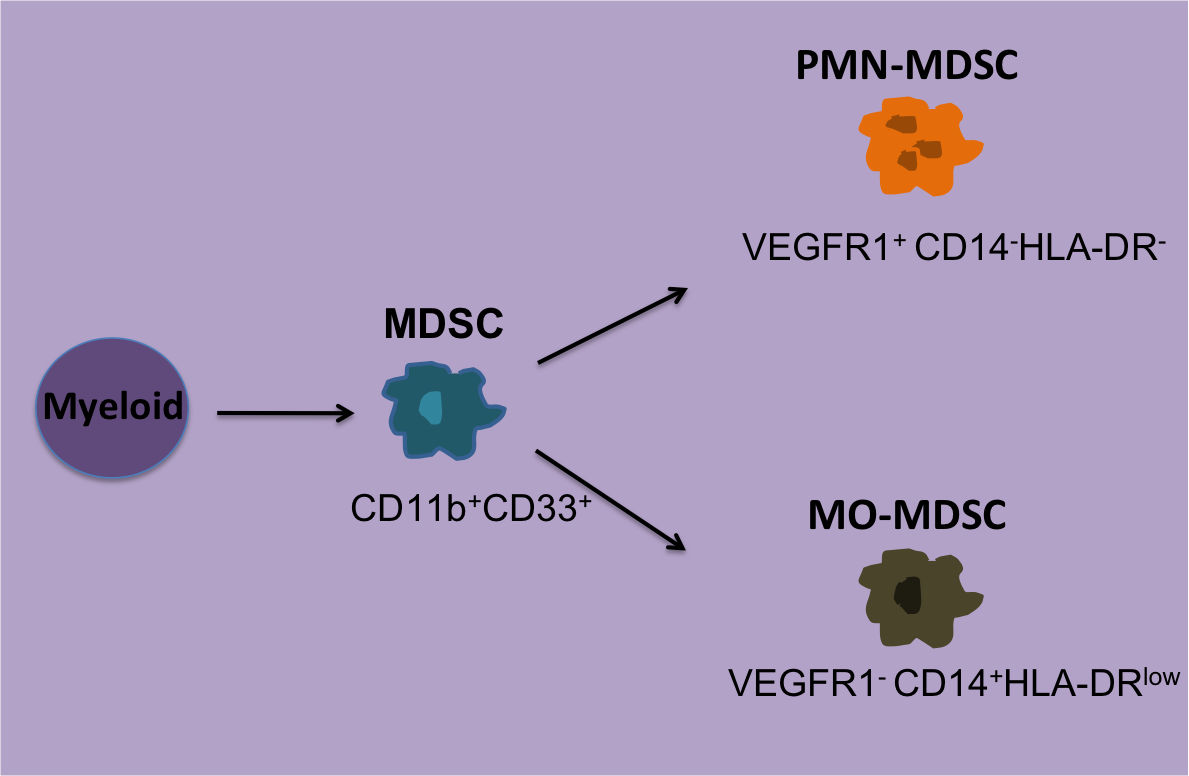
MDSCs are a heterogeneous population of immature myeloid cells that share many functions with tumor associated macrophages. Although their most noted function is a strong ability to suppress T-cell proliferation and activity. MDSCs consist of polymorphonuclear (granulocytic) and monocytic cells (PMN and MO-MDSC). PMN-MDSCs do not express HLA-DR while MO-MDSC express low levels of HLA-DR and thus both are poor antigen presenting cells. CD14 and VEGFR1 are markers that can be used to differentiate between PMN and MO-MDSCs populations. PMN-MDSC phenotype is VEGFR1+CD14– and MO-MDSCs are VEGFR1–CD14+.
Based on the pro-tumorigenic properties of MSCs they present novel targets for anti-cancer therapies. Pre-clinical studies in mouse models suggest blockade of MSC recruitment to tumors in combination with chemotherapies and anti-angiogenic treatments have beneficial effects in delaying the onset cancer cells resistance to therapy.
Further reading:
Transcriptional Profiling of the Human Monocyte-to-Macrophage Differentiation and Polarization: New Molecules and Patterns of Gene Expression
Myeloid Cells in the Tumor Microenvironment: Modulation of Tumor Angiogenesis and Tumor Inflammation