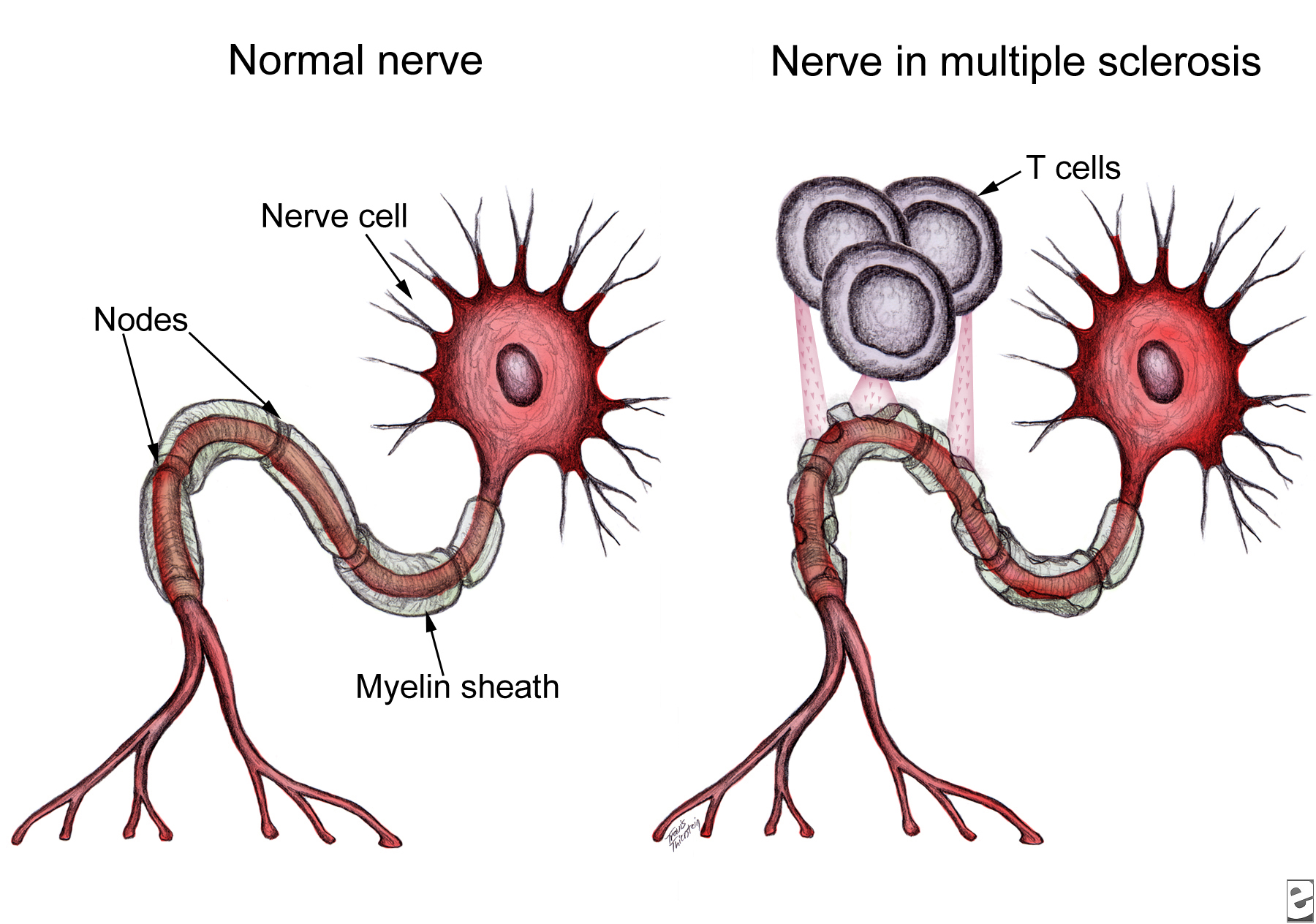
Multiple sclerosis (MS) is a degenerative inflammatory disease of the brain and the spinal cord, with its onset of symptoms occurring between the ages of 20 and 40. MS is categorized into two major forms: the most common form which accounts for 85%–90% of MS cases is relapsing-remitting MS (RRMS) whose victims usually develop secondary progressive MS (SPMS) over time. The second category, termed primary progressive MS (PPMS), accounts for approximately 10%–15% of MS cases that present with disability from the onset of the disease, progressing steadily with very little to no remissions in symptoms. It is not clear which factors are responsible for differentiating these different courses. In fact, up to date, there is little known about the underlying factors responsible for the complex heterogeneity, such as variation in immune abnormalities, observed among MS patients.
Although the etiology of MS remains unclear, it is predominantly considered to be driven by CD4+ T-cells autoreactivity to self-antigens expressed in the central nervous system (CNS), particularly to the myelin antigens. Three myelin sheath proteins that have been recognized as key autoantigens in MS include myelin basic protein (MBP), myelin oligodendrocyte protein (MOG), and proteolipid protein (PLP). Previous studies suggest epitope spreading may occur during the immune response to these three antigens in relapsing-remitting MS models. This notion is further supported by the existence of different target myelin epitopes in MS patients, which may be indicative of changes in the specificity of T-cell pathogenic response over time. These observations suggest the involvement of epitope spreading in MS, while providing a viable cause for the unfavorable efficacy reported from the several MS clinical trials that utilized a single antigen/peptidic-epitope in their therapeutic approach. In other words, previous clinical trials targeted pathogenic T-cells that are reactive against a single target antigen/epitope, which do not take into account the change of specificity in the pathogenic response overtime.
Antigen-coupled cell tolerance is a therapeutic approach aimed at antigen-specific T-cell tolerance via coupling target peptide(s) to carrier agents. In a recent study published in Science Translational Medicine, Lutterotti’s group report promising outcomes from their first-in-man MS clinical trial, demonstrating antigen-specific tolerance by autologous myelin peptide–coupled cells that utilizes a single infusion of autologous peripheral blood mononuclear cells (PBMCs) as the carrier cells. Seven myelin peptides which are believed to be key targets of autoreactive CD4+ T-cells in MS peptides (MOG1–20, MOG35–55, MBP13–32, MBP83–99, MBP111–129, MBP146–170, and PLP139–154), were chemically bound to the surface of patient-isolated PBMCs in the presence of the chemical cross-linker 1-ethyl-3-(3-dimethylaminopropyl)-carbodiimide (EDC), followed by reinfusion back to the patient. Here, Lutterotti’s group validated the safety and feasibility of this antigen-coupled cell tolerization therapeutic approach in nine MS patients. Furthermore, they reported promising tolerability resulting from their approach, since patients’ immune autoreactivity to myelin peptides were reduced by 50 to 75 percent. While all nine patients in this study displayed T-cell reactivity to at least one of the seven targeted myelin-peptides, seven were RRMS patients and two were SPMS patients.
 These results support the epitope-spreading hypothesis, which indicates that MS patients make antibodies against one or a few myelin proteins, but as the disease progresses, the autoimmune response spreads to other myelin sheath epitopes. In their recent publication, Lutterotti et al. provide sufficient evidence necessitating emphasis not only on the specific target antigens, but also on the facility to inhibit epitope spreading, preferably prior to diversification of the CD4+ T-helper cell autoreactivity.
These results support the epitope-spreading hypothesis, which indicates that MS patients make antibodies against one or a few myelin proteins, but as the disease progresses, the autoimmune response spreads to other myelin sheath epitopes. In their recent publication, Lutterotti et al. provide sufficient evidence necessitating emphasis not only on the specific target antigens, but also on the facility to inhibit epitope spreading, preferably prior to diversification of the CD4+ T-helper cell autoreactivity.
Lutterotti’s study is a significant step towards finding an effective strategy not only for treatment of MS, but also other T-cell-driven autoimmune disorders. Nonetheless, this therapeutic method must be tested on a much larger sample and geographically distinct population to demonstrate whether the efficacy reported here would be consistent in most if not all MS subtypes. The phase II of this clinical trial is planned to take place in the near future, during which more will be known about the long-term safety and efficacy of this technique. Regardless, the data presented by this study, at the very least, have set the requirement for future antigen-specific therapies to include the ability to target not only the previously activated autoreactive T-cells, but also the naïve autoreactive T-cells specific for several myelin epitopes.
Further Reading: